Description
Zetpil™ Suppository Advantage
Zetpil™ delivers nutrition via the rectum with the use of suppositories that have a unique, all- natural formulated base that has revolutionized not only this mode of delivery, but also the ability to obtain nutrients in a far more efficacious method. These suppositories are a result of combining natural ingredients with a non-irritating, rapid melting (5-6 minutes), readily absorbable (superior retention), antioxidant base material (free radical scavenging ability) and delivers the nutrients directly into the bloodstream in less than 20 minutes in a highly effective manner. Oral supplements are exposed to the hazards of stomach acid, digestive enzymes, food-related breakdown, first pass metabolism, and other bioavailability issues in the gastro-intestinal system.
Additionally, many raw materials simply have a very low bioavailability , and only achieve around 10% or less (commonly <1%) absorption rates, even if the patient is in optimal health condition. Compromised health, age, or certain conditions that result in an inability to take oral nutrients often preclude these patient populations from obtaining the necessary nutrition to stay healthy which often exacerbates chronic health issues.
Zetpil™ suppositories are a NOW solution to delivering the necessary nutrients to patients, on a daily basis, that is both easy, comfortable and cost effective. The safety and noticeable effectiveness of these products will far outweigh any initial hesitation you have regarding this mode of delivery.
What makes Zetpil™ Green Tea (EGCG) Different?
Zetpil™ EGCG suppository is unique because it represents the marriage of science and reality. By embracing the physical properties of green tea (low oral bioavailability), the researchers and scientists at Zetpil™ conceived that an alternative delivery method may increase the levels of EGCG one could achieve over a longer period of time. Based on research-proven concepts of absorption-enhancing natural compounds, excipients, therapeutically effective dosages and state-of-the-art production methods, Zetpil™ has formulated an EGCG product which is cost-effective, safe, non-competitive, avoids enzymatic breakdown in the stomach and liver and is patient-friendly. The science indicates one must absorb approximately 50mg/kg/day of body weight to achieve the predictable therapeutic benefits of green tea’s most powerful catechin- EGCG. The Zetpil™ Green Tea Extract suppository offers the health-conscious consumer the opportunity to potentially meet those levels. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3306607/
What is Green Tea?
What is Green Tea?
Green tea is made from the leaves of Camellia sinensis, and is also commonly referred to as green tea extract or the green tea catechins (GTCs) of which there are 4 main categories:
- epicatechin (EC),
- epigallocatechin (EGC),
- epicatechin gallate (ECG), and
- epigallocatechin gallate (EGCG). (http://www.howjsay.com/index.php?word=epigallocatchin-3-gallate&submit=Submit).
Of these, EGCG has been shown to be the most biologically active and contains the highest polyphenolic content accounting for 50-80% of the antioxidant properties. There are more than 17,000 peer-reviewed, scientific and medical research articles in the National Library of Medicine defining the health benefits of green tea (http://www.ncbi.nlm.nih.gov/pmc/?term=green+tea).
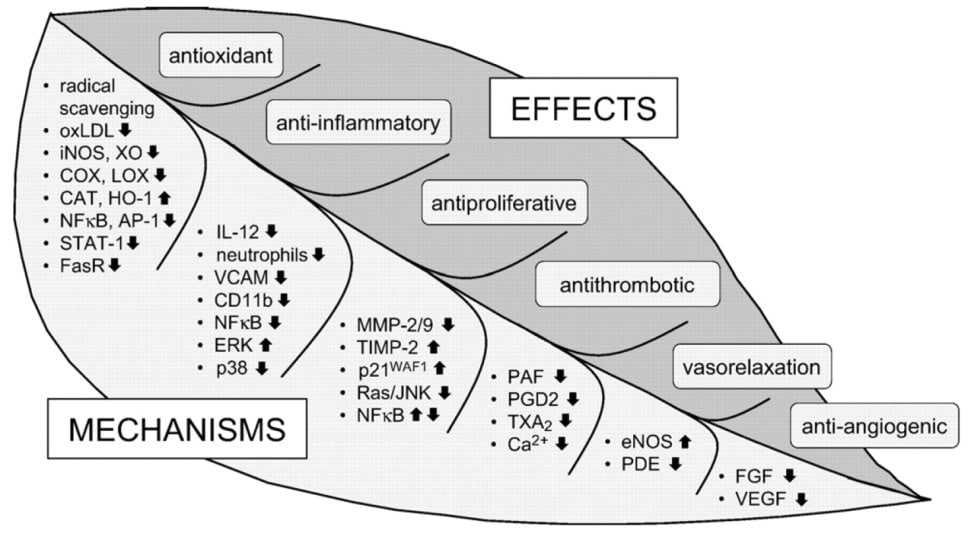
Consequently, millions of people consume green tea (beverage and/or supplement) due to its research-proven ability to act as an anti-inflammatory, anti-arthritic, antioxidant, anti-bacterial, neuro-protectant, and cholesterol-lowering compound. In fact, green tea is the second most consumed beverage in the world next to water.
Research has shown that Green Tea may be beneficial for:
- improving blood pressure;*
- reducing LDL (bad cholesterol) and increasing HDL (good cholesterol) levels;*
- decreasing the risk of developing atherosclerosis through its anti-inflammatory properties;*
- regulating healthy glucose levels as well as carbohydrate metabolism (weight loss and anti-diabetic effects);*
- anti-inflammatory and antioxidant properties;*
- proper immune function;*
- reducing the risk of neurodegenerative diseases (i.e. Alzheimer’s);*
- maintaining a healthy cardiovascular system; and*
- preventing certain types of cancer through its free radical scavenging ability.*
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.*
Problems with Beverages & Oral Ingestion
Green Tea Beverages or Oral Supplements cannot reach Therapeutic Dosages for Serious Health Issues
Millions of people consume green tea daily for its health benefits, and while certainly a health-conscious choice over other beverages, drinking green tea or taking oral supplements will not afford you the necessary therapeutic dosages required in disease crises.
- A 2013 article published in Current Pharmaceutical Design explains that “in humans, plasma bioavailability of green tea catechins is very low. After the administration of 697 mg of green tea, plasma EGCG was only 0.07%” of the ingested dose. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4055352/
- The International Journal of Molecular Science published an article in 2011 confirming that “most of ingested EGCG apparently does not get into the blood, since absorption takes place in the small gut and substantial quantities pass from the small to the large intestine where it undergoes further degradation by the action of local microbiota.”http://www.mdpi.com/1422-0067/12/9/5592
- In 2012, the European Journal of Nutrition made a breakthrough discovery about food chemistry interactions that occur upon oral consumption of green tea. They stated that “plasma concentrations of EGCG were significantly lower after the consumption of green tea with dietary proteins (casein, soy protein, or skim milk) than after green tea alone. The effects of soy protein were similar to those observed after the addition of milk.” http://www.ncbi.nlm.nih.gov/pubmed/22366739
Therapeutic Potential
Therapeutic Potential of Green Tea Extract (EGCG)
Arthritis
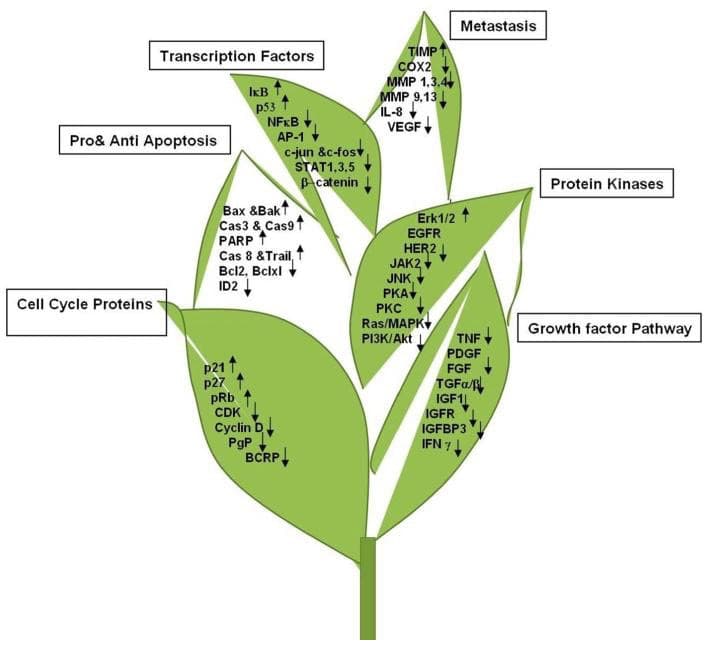
“Nuclear factor kappa Beta (NF-κB) acts as a controlling switch for the regulation of inflammation, innate immunity, and arthritis. We have previously shown that Epigallocatechin-3-gallate (EGCG) inhibits NF-κB activity and can alter the activity of inflammatory cells involved in arthritis. EGCG may also be helpful in protecting joints from oxidative damage associated with chronic inflammation in arthritis.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3146294/
Cardiovascular Disease
“Consumption of tea is increasingly being shown to be associated with enhanced cardiovascular and metabolic health. In addition, green tea intake decreases the absorption of triglycerides and cholesterol and these findings are in accordance with the fact that (green tea) increases excretion of fat.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4055352/
Cancer
“GTE (Green Tea Extract) has a strong effect on the functional properties of metastatic cancer cells in live, clinically obtained patient samples. Unlike other chemotherapeutic drugs, Green Tea Extract (GTE) was selective on cancer cells but not the non-cancer cells in the same specimen.” http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3151463/pdf/nihms308363.pdf
http://cardiovascres.oxfordjournals.org/content/73/2/348.long
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3303150/figure/F2/
Supplement Facts
Supplement Facts
| Serving Size: 1 suppository
Servings per Container: 30 |
|
| Green tea Extract 94 SR (Sustained Release) contains the following ingredients:
400 mgs of epigallocatechin 3-gallate 94% EGCG (426mgs) from Taiyo Sunphenon green tea extract |
|
| Suppository Base: The base is a proprietary combination of excipients plant and fruit butters, medium chain triglycerides, phospholipids, lecithin and arrow root starch. | |
This product is contraindicated in individuals with a known hypersensitivity to phospholipids, or any of the listed ingredients.
Due to the nature of the product suppositories cannot be returned or exchanged.
If you chose to use this product as a rectal suppository, it is highly recommended you attempt to clear your bowels prior to inserting it to avoid premature evacuation. To facilitate insertion it is recommended you moisten the product by passing it under a stream of cool water. When used as a suppository, this product requires approximately 5 minutes to dissolve and begin to be absorbed. However in order to maximize the therapeutic effect of94% EGCg (epigallocatechin 3-gallate) the suppository has been formulated to allow a sustained release of the EGCg over a 4-5 hour period of time for complete absorption of the ingredients. Although not required, if inserted while lying on one’s side, consider remaining in the supine position for 1-3 minutes following insertion.
WARNINGS:
Although safe for children, this product should be kept out of the reach of children.
Do not use if the suppositories are open or damaged.
As with any nutrient if you are pregnant or breastfeeding, seek the advice of a health care provider before using this product.
Individuals that have chosen to take this product as part of a personal nutrient program have overwhelmingly chosen to utilize this product as a rectal suppository. However, this product can also be taken safely by mouth. If one chooses to take this product orally, then this product may be considered a dietary supplement in accordance with current FDA guidelines.
Disclaimer: These statements have not been evaluated by the FDA. This product is a Federal Food, Drug, and Cosmetic Act compliant combination of naturally derived FDA GRAS (Generally regarded As Safe) herbal, nutraceutical, vitamins, minerals and amino acids products legally sold in the USA. In accordance with FDA regulations, when taken orally, this product may be considered a dietary supplement as defined by section 201(ff) of the Act, 21 U.S.C. § 321(ff). Also, this product consists ONLY of a combination of naturally derived FDA GRAS (Generally regarded As Safe) herbal, nutraceutical, vitamins, minerals and amino acids products legally sold in the USA. Therefore, this product cannot be considered a drug as defined in section 201(g)(1) of the Act, 21 U.S.C. § 321(g)(1). This product is not intended to affect the structure or function of the body and is not intended for use in the diagnosis, cure, mitigation, treatment or prevention of disease in man. Any nutritional suggestions and research regarding this product are provided for informational purposes only, and are not intended to diagnose, treat, cure or prevent disease nor should any information provided be used as a substitute for sound medical advice.
References
References
There are more than 17,000 free peer-reviewed scholarly articles about green tea (EGCG) at the U.S. National Library of Medicine (www.ncbi.com). We encourage anyone looking for scientific and medical information to follow this link. http://www.ncbi.nlm.nih.gov/pmc/?term=green%20tea
Tea and Health: Studies in Humans
Curr Pharm Des 2013; 19(34): 6141–6147
Naghma Khan, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4055352/
Green tea polyphenol epigallocatechin-3-gallate: inflammation and arthritis
Life Sci Jun 19, 2010; 86(25-26): 907–918
Rashmi Singh, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3146294/
Epigallocatechin-3-gallate (EGCG) for Clinical Trials: More Pitfalls than Promises?
Derliz Mereles and Werner Hunstein
Int. J. Mol. Sci. 2011, 12(9), 5592-5603
http://www.mdpi.com/1422-0067/12/9/5592
Green tea supplementation increases glutathione and plasma antioxidant capacity in adults with the metabolic syndrome
Nutr Res Mar 2013; 33(3): 180–187
Arpita Basu, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3603270/
Green tea: A boon for periodontal and general health
J Indian Soc Periodontol. 2012 Apr-Jun; 16(2): 161–167
Anirban Chatterjee, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3459493/
Antioxidant effects of green tea
Mol Nutr Food Res Jun 2011; 55(6): 844–854
SARAH C. FORESTER, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3679539/
Green tea supplementation increases glutathione and plasma antioxidant capacity in adults with the metabolic syndrome
Nutr Res Mar 2013; 33(3): 180–187
Arpita Basu, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3603270/
Tea polyphenols for health promotion
Life Sci Jul 26, 2007; 81(7): 519–533
Naghma Khan, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3220617/
Green Tea and Bone metabolism
Nutr Res Jul 2009; 29(7): 437–456
Chwan-Li Shen, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2754215/
Tea and Health: Studies in Humans
Curr Pharm Des 2013; 19(34): 6141–6147
Naghma Khan , et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4055352/
Effect of 2 month controlled green tea intervention on lipoprotein cholesterol, glucose, and hormone levels in healthy postmenopausal women
Cancer Prev Res (Phila) Mar 2012; 5(3): 393–402
Anna H. Wu, et al
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3777853/
Green tea extract selectively targets nanomechanics of live metastatic cancer cells
Cross SE, et al
Nanotechnology 2011 May 27; 22(21):215101
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3151463/pdf/nihms308363.pdf